Every year in the United States, more than 48,000 dogs are confined in laboratory facilities — the overwhelming majority of them beagles. At the same time, monkeys are captured, transported, and shipped into biomedical research environments where stress, confinement, and invasive experimentation define their daily existence.
This is not simply an animal welfare issue.
It is a sustainability issue.
It is a scientific integrity issue.
It is a regulatory modernization issue.
At Sustainable Action Now (SAN), our commitment to sustainability extends beyond climate and energy systems. True sustainability includes ethical science, responsible innovation, and the transition toward human-relevant research methodologies that reduce suffering while increasing accuracy.
The era of default animal testing is ending. And it must.
The Reality Behind Laboratory Doors
Beagles are frequently used in toxicology studies because of their docile temperament and manageable size. Monkeys are used because of their genetic proximity to humans. Yet similarity does not equal reliability — and captivity does not equal consent.
Animals in laboratories experience:
- Invasive procedures
- Forced drug exposure
- Surgical interventions
- Long-term confinement
- Social isolation
- Psychological stress
They cannot opt out. They cannot provide informed consent. They cannot communicate distress in human language.
And increasingly, science shows they do not reliably predict human outcomes.
The Scientific Case Against Animal Testing
1. Scientific Unreliability
Between 90% and 96% of drugs that pass animal testing fail in human clinical trials — often because of unforeseen toxicity or lack of effectiveness. That is not a marginal failure rate. It is systemic inefficiency.
If nearly all animal-tested drugs fail in humans, the model is not merely imperfect. It is structurally flawed.
2. Biological Incompatibility
Animals do not metabolize drugs the way humans do. Differences in physiology, organ function, immune response, and disease progression fundamentally limit predictive accuracy.
Examples are instructive:
- Penicillin is fatal to guinea pigs but life-saving for humans.
- Aspirin is toxic to cats yet widely used in human medicine.
These inconsistencies underscore a central truth: cross-species extrapolation is not precision science.
3. Economic Inefficiency
Animal facilities are costly to operate. Housing, feeding, veterinary care, regulatory compliance, and infrastructure add enormous overhead.
Completing animal studies for a single pesticide can take up to a decade and cost millions of dollars. When misleading data derails promising treatments — or allows harmful drugs to advance — those costs multiply.
4. Ethical Imperative
Animals are sentient beings capable of experiencing pain, fear, and psychological distress. The ethical argument is not abstract — it is rooted in cognitive science and behavioral evidence.
Sustainability requires systems that minimize harm where alternatives exist.
And alternatives now exist.
The Rise of New Approach Methodologies (NAMs)
Effective Alternatives (NAMs)
These methods prioritize human biology and are increasingly supported by regulatory bodies like the FDA.
| Alternative | Description | Key Advantage |
|---|---|---|
| Organs-on-a-Chip | Microchips lined with living human cells that mimic organ functions (lung, liver, heart). | Replicates human physiology more accurately than animal models. |
| In Silico (AI & Modeling) | Sophisticated computer simulations and AI that predict how drugs will interact with the human body. | Extremely fast; can process complex data in minutes. |
| Human Organoids | Lab-grown, 3D “mini-organs” derived from human stem cells. | Provides a direct window into human-specific disease mechanisms. |
| In Vitro Testing | Testing chemicals on human cell cultures or lab-grown human skin (e.g., EpiDerm). | Produces more relevant results for human skin sensitivity than rabbits. |
| Microdosing | Administering extremely small, safe doses of a drug to human volunteers for tracking. | Provides actual human metabolic data at the earliest stage of research. |
| 3D Bioprinting | Using “bio-inks” to print human-like tissues for drug screening. | Reduces drug failure rates by testing on human-specific tissue structures. |
The scientific community is rapidly transitioning toward New Approach Methodologies (NAMs) — human-based research models that are faster, cheaper, and more predictive.
Regulatory frameworks are evolving accordingly. The U.S. Food and Drug Administration is increasingly supportive of non-animal testing data, particularly following passage of the FDA Modernization Act 2.0, which removed mandatory animal testing requirements for drug approvals and encouraged validated alternatives.
This legislative shift is not symbolic. It is structural.
Below are the most transformative NAM technologies reshaping biomedical research.
Organs-on-a-Chip: Microengineering Human Physiology
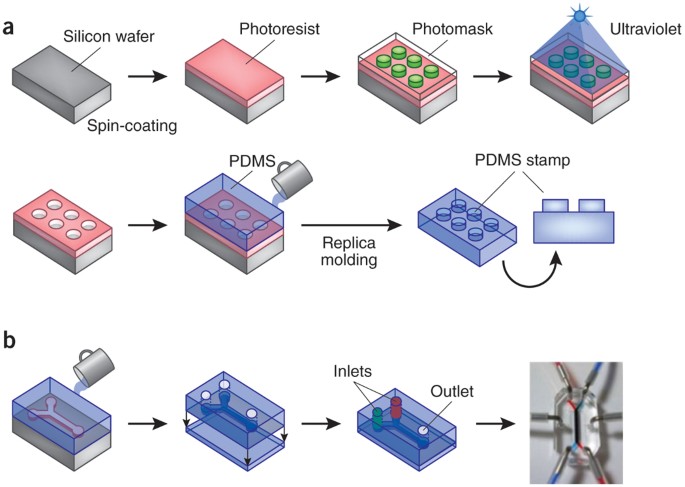
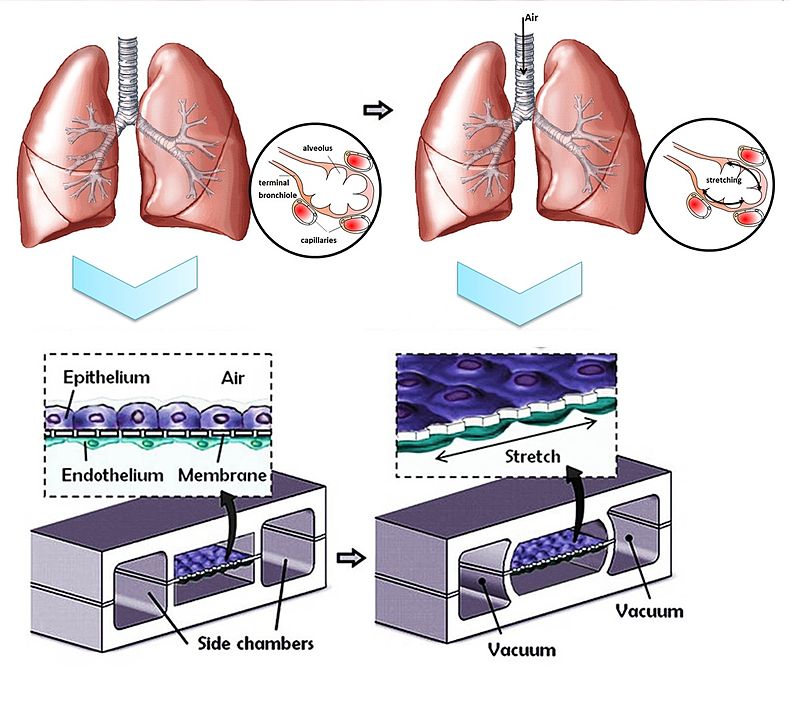

Organs-on-a-chip are microfluidic devices lined with living human cells that replicate organ-level function — including lung expansion, liver metabolism, and cardiac contraction.
Key Advantages:
- Mimic human-specific responses
- Allow real-time toxicity monitoring
- Reduce cross-species error
- Enable precision dosing studies
These systems simulate human physiology with far greater relevance than rodent or primate models.
Human Organoids: Miniature Living Systems
Human organoids are lab-grown three-dimensional “mini-organs” derived from stem cells. They replicate structural and functional features of organs such as the brain, liver, and intestines.
Why It Matters:
- Direct human disease modeling
- Personalized medicine potential
- Genetic specificity
- Improved cancer and neurological research outcomes
Organoids eliminate species translation gaps while preserving biological complexity.
In Silico Modeling: AI-Driven Drug Prediction
Advanced computational models now simulate drug interactions using artificial intelligence and big data integration.
Advantages:
- Process complex pharmacokinetic data in minutes
- Reduce trial-and-error experimentation
- Identify toxicity risks before human exposure
- Cut development timelines dramatically
In silico modeling is not theoretical. It is already embedded in pharmaceutical pipelines.
In Vitro Testing & Lab-Grown Human Tissue
Human cell cultures and reconstructed human skin models (such as EpiDerm-type systems) provide accurate toxicity and sensitivity data without relying on rabbits or rodents.
These models:
- Deliver direct human relevance
- Improve dermatological testing accuracy
- Eliminate unnecessary animal suffering
Microdosing and Early Human Data
Microdosing involves administering extremely small, safe doses of a drug to human volunteers while tracking metabolic response using advanced imaging technologies.
Impact:
- Provides real human metabolic data early
- Identifies viability before large-scale trials
- Reduces dependency on speculative animal models
This is evidence-based modernization.
3D Bioprinting: Precision Tissue Engineering
3D bioprinting uses bio-inks composed of living cells to print human-like tissues for screening drugs.
This technology:
- Replicates tissue architecture
- Enhances screening accuracy
- Reduces late-stage drug failures
- Improves predictive toxicology
As bioprinting scales, reliance on animal models becomes increasingly obsolete.
Why Monkeys Should Not Be Shipped to Labs
Primates are cognitively complex, socially bonded, and emotionally responsive. Removing them from natural environments, transporting them across borders, and confining them for invasive research introduces extreme psychological stress.
High cortisol levels, self-harming behaviors, immune suppression, and trauma are documented outcomes of prolonged laboratory confinement.
When scientifically superior alternatives exist, subjecting sentient primates to invasive testing becomes not only ethically questionable but scientifically unnecessary.
Sustainability demands we retire outdated practices.
Economic Sustainability and Innovation
Modernizing research methods is not anti-science. It is pro-innovation.
Benefits of transitioning away from animal testing include:
- Reduced development timelines
- Lower operational costs
- Improved predictive accuracy
- Reduced legal liability
- Increased public trust
The biotech sector is already pivoting toward human-centric models because efficiency and ethics align.
This is what sustainable innovation looks like.
Policy Momentum and Regulatory Reform
The regulatory landscape is shifting. The FDA Modernization Act 2.0 and subsequent updates formally acknowledge non-animal testing as acceptable in drug development pathways.
This legislative evolution signals:
- Institutional recognition of scientific limitations
- Federal openness to validated alternatives
- Acceleration of NAM integration
The policy framework now supports transformation.
The remaining question is implementation speed.
Sustainable Action Now: The Ethical Innovation Mandate
At SAN, we advocate for:
- Full federal funding for NAM development
- Accelerated regulatory validation of non-animal methods
- Transparent reporting on laboratory animal use
- Ethical procurement standards
- Corporate accountability in pharmaceutical testing
Sustainability is not solely environmental. It is systemic. It requires updating legacy structures when better models emerge.
The data is clear. The technology exists. The regulatory door is open.
Take Action
Support policies that fund human-based research technologies.
Advocate for full adoption of New Approach Methodologies.
Encourage companies to publicly commit to non-animal testing pathways.
Engage lawmakers on expanding FDA modernization efforts.
Every year, tens of thousands of dogs and countless primates endure laboratory confinement.
Science no longer needs to rely on their suffering.
Sustainable systems evolve.
Ethical innovation replaces outdated practice.
And responsible action moves us forward.
The future of research is human-relevant, technology-driven, and cruelty-free.
It is time to accelerate the transition.



